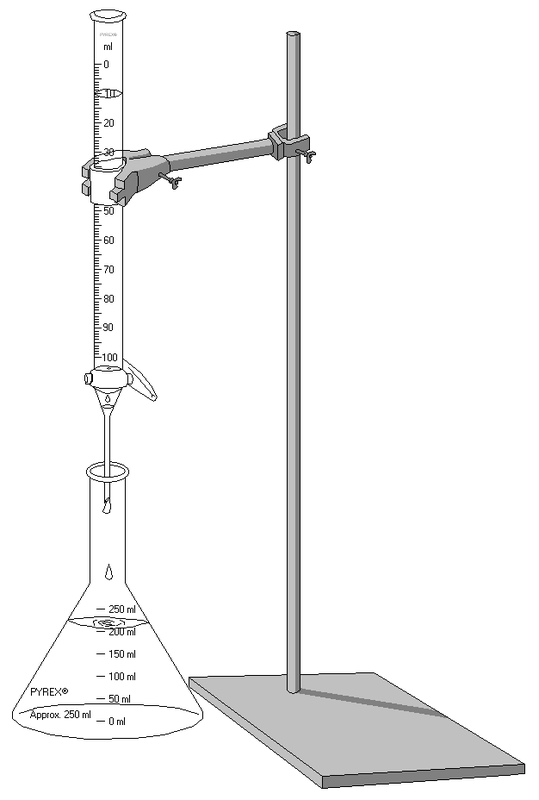
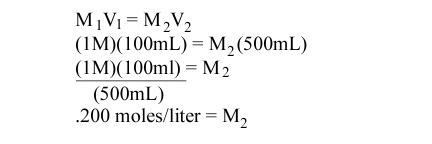Here is a youtube video explaining on how to do a titration
What is a titration?
A titration is a process of neutralizing that slowly adds one solution (titrant) to another solution until it reaches neutralization.
|
Rough titration
Rough estimate to what the titration is |
Exact titration
The exact or very accurate to what the titration is |
Sample Procedure
|
Molarity in titration
If the molarity of the solution is unknown, you use the formula M1V1 = M2V2.
Click here if you do not know anything about molarity.
So, if you are trying to find the molarity of 10 mL of HCL and you are mixing this with 10 mL of 1 mol sodium hydroxide, you do:
M(10)=1(10)
this means the molarity is 1.
If the molarity of the solution is unknown, you use the formula M1V1 = M2V2.
Click here if you do not know anything about molarity.
So, if you are trying to find the molarity of 10 mL of HCL and you are mixing this with 10 mL of 1 mol sodium hydroxide, you do:
M(10)=1(10)
this means the molarity is 1.
Good job! Click here to take the final test! Good luck!