Buffers
What is a buffer?
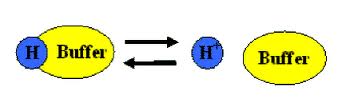
a solution that resists changes in pH when acid or alkali is added to it. This means that the buffer does not change pH with the proper acid and base combination
a solution that resists changes in pH when acid or alkali is added to it. This means that the buffer does not change pH with the proper acid and base combination
What are buffers composed of?
Buffers consistent of a weak conjugate acid-base pair. This means either a weak acid and its conjugate base, or a weak base and its conjugate acid.
Buffers consistent of a weak conjugate acid-base pair. This means either a weak acid and its conjugate base, or a weak base and its conjugate acid.
Uses for buffers?
Click below to learn what a titration is!