Strength and Concentration
Strength of an acid
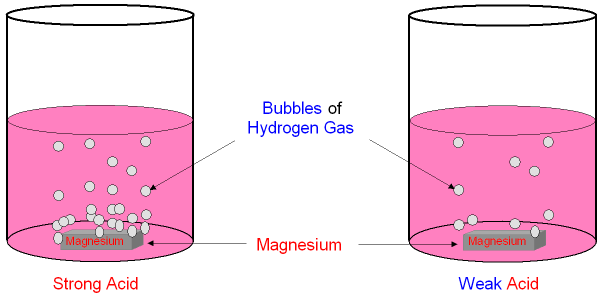
Strength of an acid is the amount it disassociates to form hydrogen ions
A strong acid completely dissociates to from hydrogen ions while a weak acid will only partially disassociates to form hydrogen ions
Strength of an acid is the amount it disassociates to form hydrogen ions
A strong acid completely dissociates to from hydrogen ions while a weak acid will only partially disassociates to form hydrogen ions
Strength of a base
The concentration of the base relates to the strength of the base. The strength of a base is equal to the amount the base disassociates from [OH-].
The concentration of the base relates to the strength of the base. The strength of a base is equal to the amount the base disassociates from [OH-].
pH levels
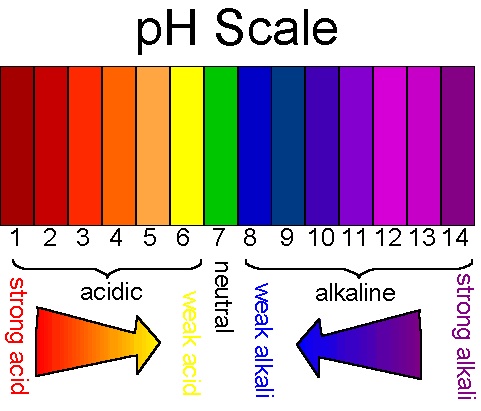
pH of a strong acid: 1-3
pH of a weak acid: 4-6
pH of a weak base: 8-10
pH of a weak base: 11-14
pH of a strong acid: 1-3
pH of a weak acid: 4-6
pH of a weak base: 8-10
pH of a weak base: 11-14
Concentration
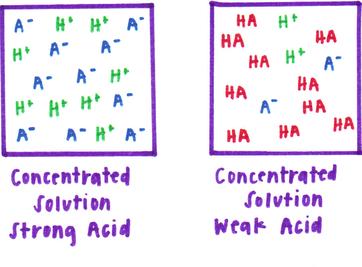
The concentration of a liquid is the amount of the acid/base per liter. A concentrated solution is one that has a large amount of acid/base per liter while a diluted solution is one that contains a smaller amount of a acid/base per liter.
The concentration of a liquid is the amount of the acid/base per liter. A concentrated solution is one that has a large amount of acid/base per liter while a diluted solution is one that contains a smaller amount of a acid/base per liter.
Click below to learn about what conjugate acids and bases are