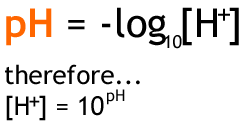pH, pOH, [H+] and [OH-] calculations
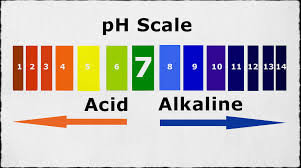
The pH and pOH is what determines the strength of an acid. As I said before, the pH scale ranges from 1-14. The closer to 0 the solution is, the more acidic said solution is. The closer the pH of the solution is 14, the more basic said solution is. However, when the solution has a pH of 7, then the solution is neutral
Calculations
When you are given the [H+], [OH-], pH or pOH, you can calculate the rest of the values
Sample calculation
Lets say that the [H+] value is .0001
To calculate the pH, you use the formula -log{H+} = pH
So, -log(.0001) = 4
This means the pH of the solution is 4
This calculation is the same for pOH,
If the [OH-] value is .0001, than using the -log equation, you can figure out the pOH is 4
Now, lets say you have the pH value, you use this equation to find the [H+]
10^(-pH)
So if the pH is 4, using this equation, 10^(-4), you know that the [H+] is 10^-4 or .0001
This goes for if you have the pOH and you need to use the equation 10^(-OH-)
Also, to calculate the pH or pOH when given the opposite value, pH + pOH = 14. So it is easy to figure out the pH or pOH when given the opposite value
When you are given the [H+], [OH-], pH or pOH, you can calculate the rest of the values
Sample calculation
Lets say that the [H+] value is .0001
To calculate the pH, you use the formula -log{H+} = pH
So, -log(.0001) = 4
This means the pH of the solution is 4
This calculation is the same for pOH,
If the [OH-] value is .0001, than using the -log equation, you can figure out the pOH is 4
Now, lets say you have the pH value, you use this equation to find the [H+]
10^(-pH)
So if the pH is 4, using this equation, 10^(-4), you know that the [H+] is 10^-4 or .0001
This goes for if you have the pOH and you need to use the equation 10^(-OH-)
Also, to calculate the pH or pOH when given the opposite value, pH + pOH = 14. So it is easy to figure out the pH or pOH when given the opposite value
Click below to take the quiz!