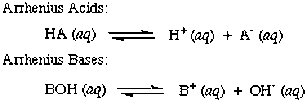Definitions and Theories
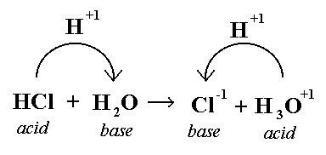
Bronsted Lowry Theory
The Bronsted Lowry Theory states that an acid donates hydrogen ion protons, while the bases receive the hydrogen ion protons.
The Bronsted Lowry Theory states that an acid donates hydrogen ion protons, while the bases receive the hydrogen ion protons.
Arrhenius Theory
When acids dissolve in water they produce hydrogen (H+) ions and when bases are dissolved in water they produce hydroxide ions (OH-).
When acids dissolve in water they produce hydrogen (H+) ions and when bases are dissolved in water they produce hydroxide ions (OH-).
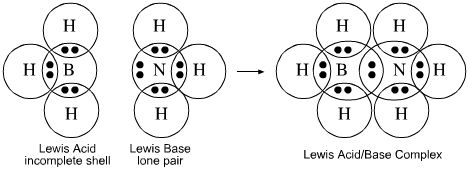
Lewis Theory
Acids are electron pair acceptors while bases are electron pair donors.
Acids are electron pair acceptors while bases are electron pair donors.
Definition of Acid and Base
Acid: An acid is a proton (H+) donor, also accepts electron pairs and produce hydrogen ions (H+)
Base: A base receives protons, also are electron pair donors and finally produce hydroxide ions (OH-)
Acid: An acid is a proton (H+) donor, also accepts electron pairs and produce hydrogen ions (H+)
Base: A base receives protons, also are electron pair donors and finally produce hydroxide ions (OH-)
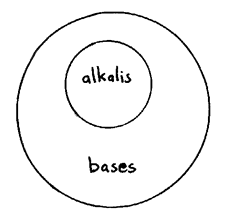
Alkali
An alkali is a soluble base. All alkalies are bases but not all bases are alkalies
An alkali is a soluble base. All alkalies are bases but not all bases are alkalies
Click the button below to continue the lesson